Treatment for Severe Emphysema
The vision of Pulmonx is to be a global leader in bronchoscopic treatment for emphysema. The Zephyr® Valve System has clinically-proven results, reliability, patient selection tools, and ease of use.
Zephyr Valve
How it Works
Minimally Invasive Bronchoscopic Procedure
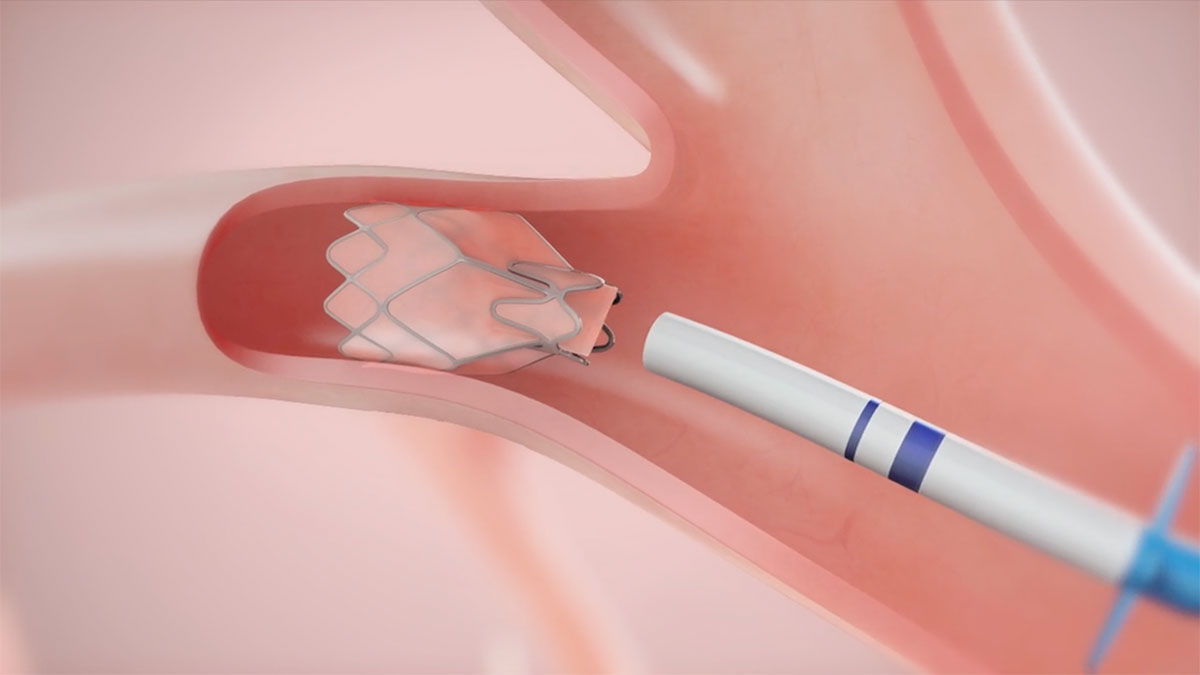
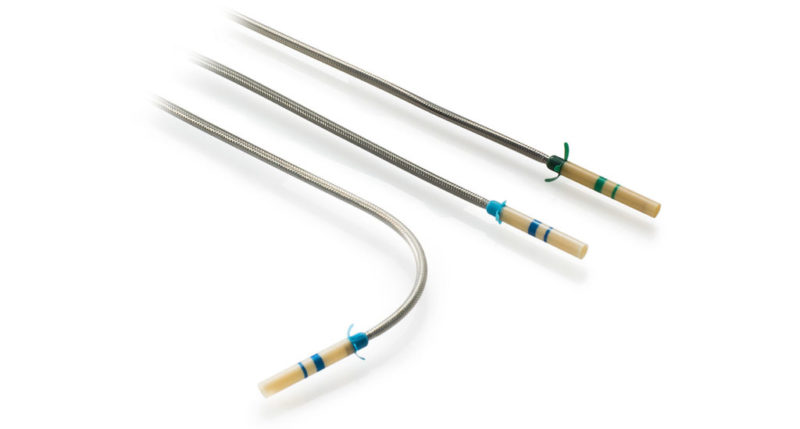
The Zephyr Endobronchial Valve was granted a “breakthrough medical status”20 by the FDA and is indicated for bronchoscopic treatment of patients with hyperinflation associated with severe emphysema in regions of the lung that have little to no collateral ventilation (CV). The Zephyr Valve is an implantable device used to occlude all airways feeding the hyperinflated lobe of a lung that is most diseased with emphysema.
Trapped air escapes through the Zephyr Valves until the lobe volume is reduced. The remaining lobes are then able to expand more fully and work more efficiently, reducing pressure on the diaphragm and improving overall lung function.
- 30 to 60-minute procedure
- Typically, 3 to 5 valves placed to completely occlude the lobe
- Can be removed or replaced if needed
- 3-night stay in the hospital
Complications of the Zephyr Endobronchial Valve treatment can include, but are not limited to, pneumothorax, worsening of COPD symptoms, hemoptysis, pneumonia, dyspnea and, in rare cases, death.
Design Features
Demonstrated to Have Consistent Patient Benefits
The Zephyr Valve is specially designed to adapt to the dynamic conditions of the airway.
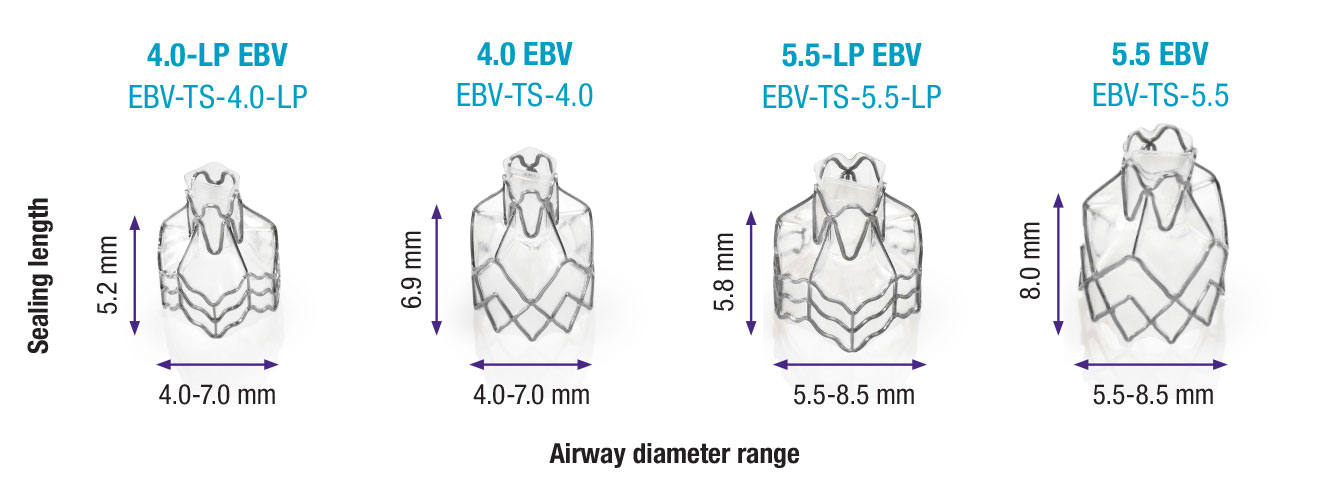
Broad Range of Airway Sizes
Simple and Flexible Sizing
- Simple and flexible sizing to accommodate a full range of airway sizing
- Successful occlusion can be accomplished for a wide range of airway shapes
Zephyr Endobronchial Delivery Catheter (EDC)
Technology for Precise Patient Selection
Appropriate patient selection is critical to the success of Zephyr Valve treatment. Clinical trial data have helped define the successful patient profile.17
Prior to treatment, physicians perform a clinical workup, including lung function and hyperinflation testing. In addition, Pulmonx offers assessment tools that precisely identify patients who are most likely to benefit from Zephyr Valve treatment.
Assess with Certainty
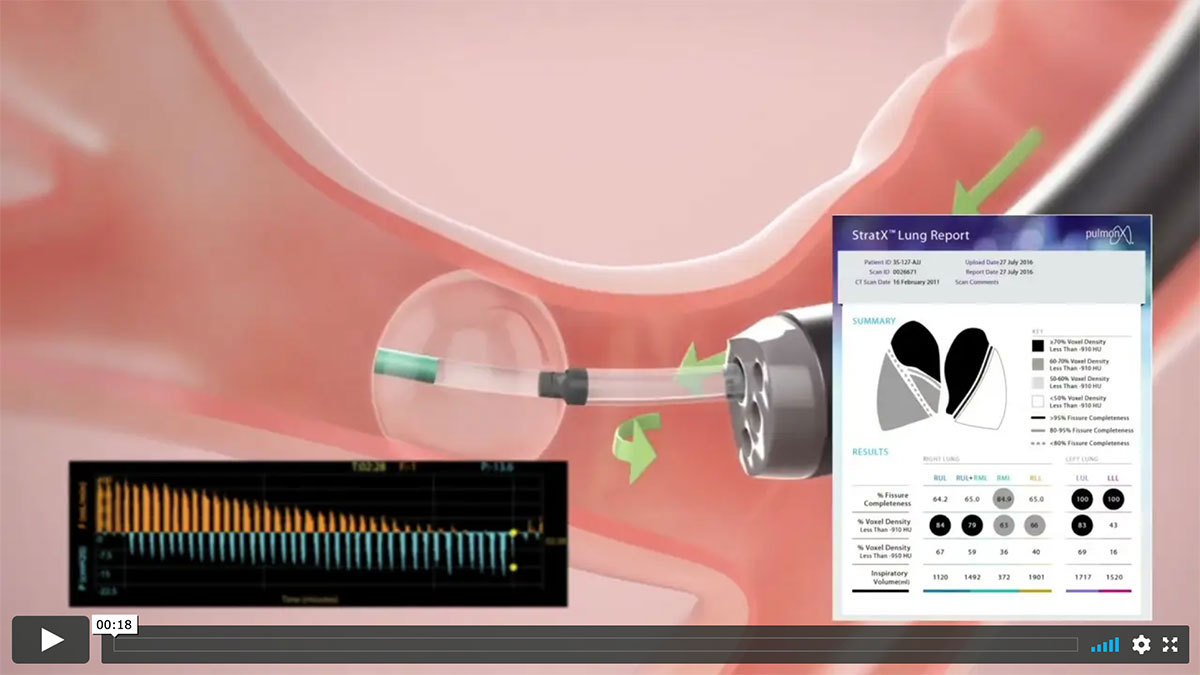
StratX® Lung Analysis Platform
The StratX® Lung Analysis Platform is a cloud-based quantitative CT analysis service that provides information on:
- Emphysema destruction
- Fissure completeness
- Lobar volumes
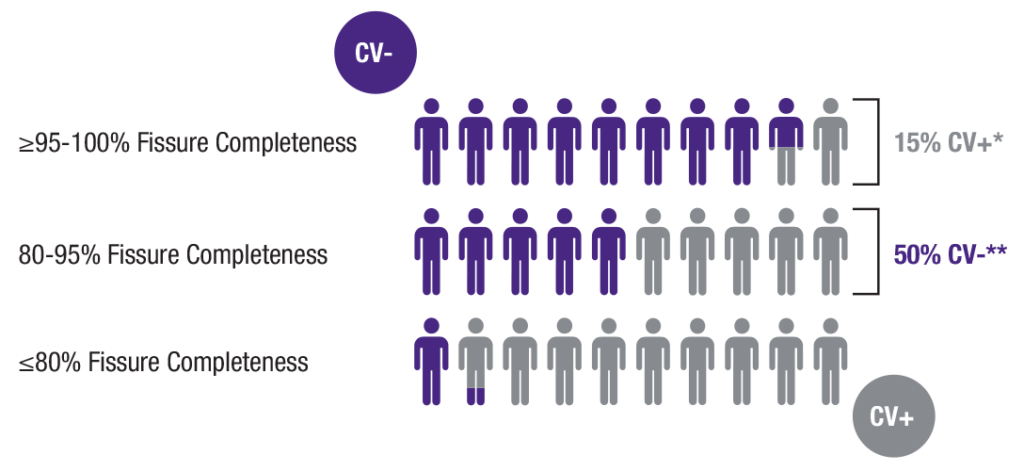
- <80% fissure completeness score considered positive for collateral ventilation (CV+)
- >80% fissure completeness score likely to be negative for collateral ventilation (CV-)
- CV status in these patients should be confirmed with the Chartis® Pulmonary Assessment System
Treat with Confidence
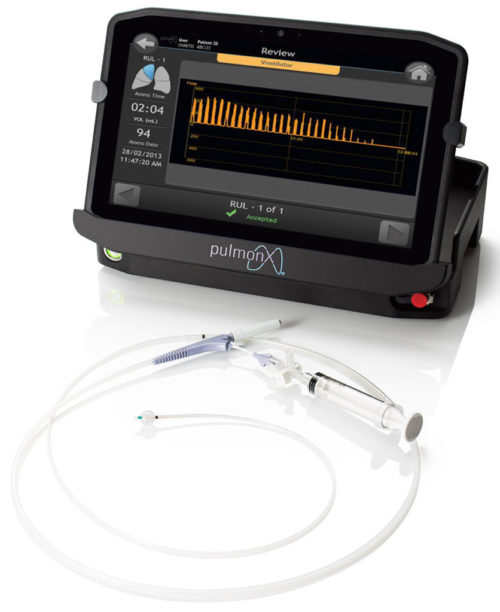
Chartis® Pulmonary Assessment System
The Chartis® Pulmonary Assessment System is a proprietary balloon catheter and console with flow and pressure sensors used to assess the presence of collateral ventilation (CV). It is a physiological measure of collateral ventilation.
More Than 3 in 4 Patients Would Select a Treatment with the Clinical Benefits and Risk Profile of the Zephyr Valve Over Current Treatment.8
A detailed survey of 294 severe emphysema patients evaluated how they balance potential risks and benefits. The study concluded that patients with severe emphysema value access to an interventional treatment that offers benefits above and beyond their current medical management, despite the risks associated with these treatments.