The benefits are comparable to those seen with LVRS (lung volume reduction surgery) but with a reduction in post-procedure morbidity.”1
Published in The American Journal of Respiratory and Critical Care Medicine1
A multicenter, multinational randomized controlled trial of Zephyr® Endobronchial Valves in patients with heterogenous emphysema and little to no collateral ventilation.
Methods & Endpoints
- First multicenter to evaluate effectiveness and safety of Zephyr Endobronchial Valves in patients with little to no collateral ventilation (CV) out to 12 months
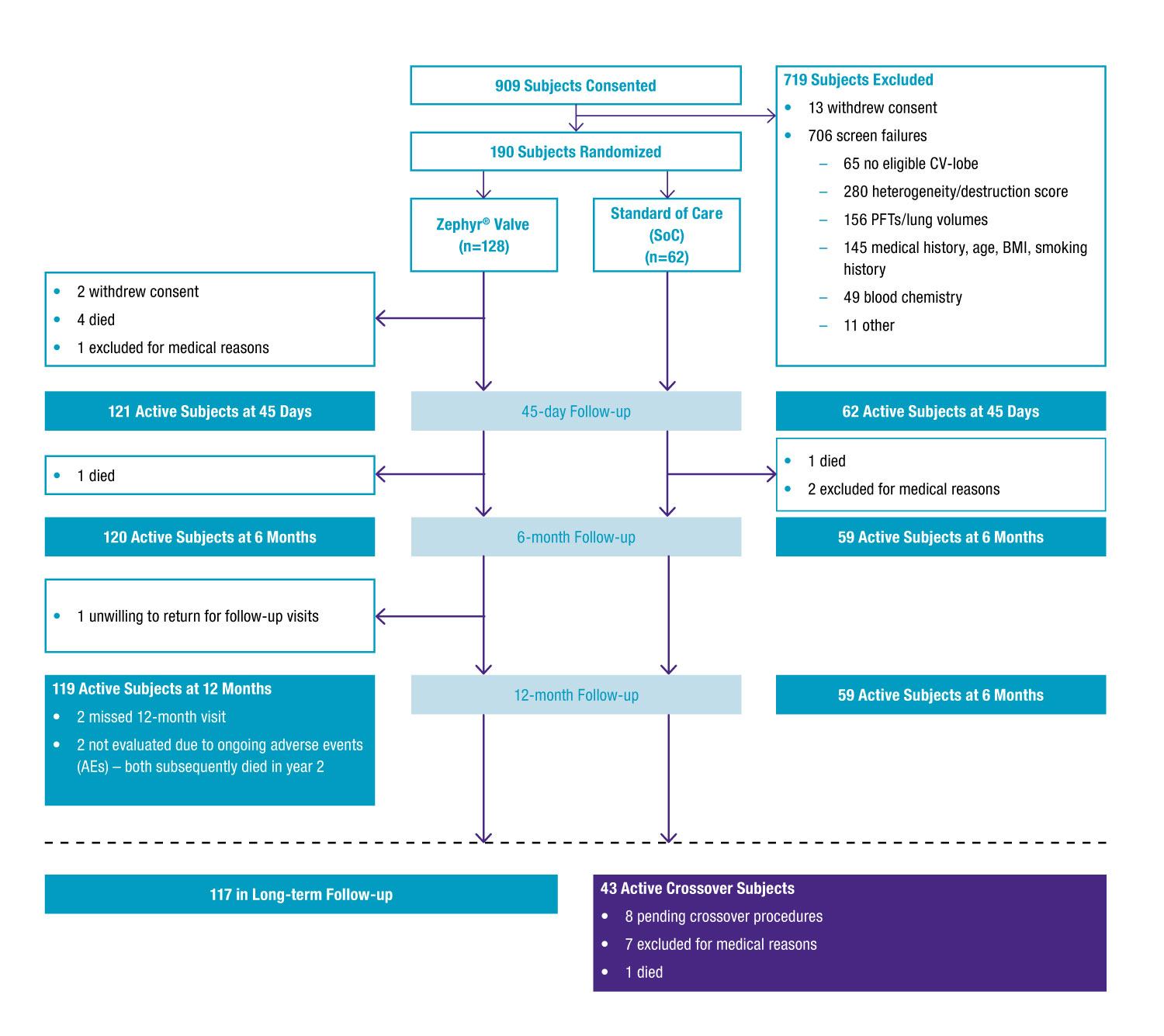
- 190 subjects with hyperinflation (mean baseline RV, 225% pred.; FEV1, 27% pred.; DLCO, 34% pred.) randomized 2:1 (128 Zephyr Valve: 62 SoC)
Study Design
Results in ITT Population
Primary Endpoint Responder Analysis
Percent of Subjects with FEV1 Change of ≥15%
Other Responder Analysis
Proportion of Patients Achieving MCID
Secondary Endpoints
Change in St. George’s Resp Questionnaire
Change in FEV1
Change in 6MWD
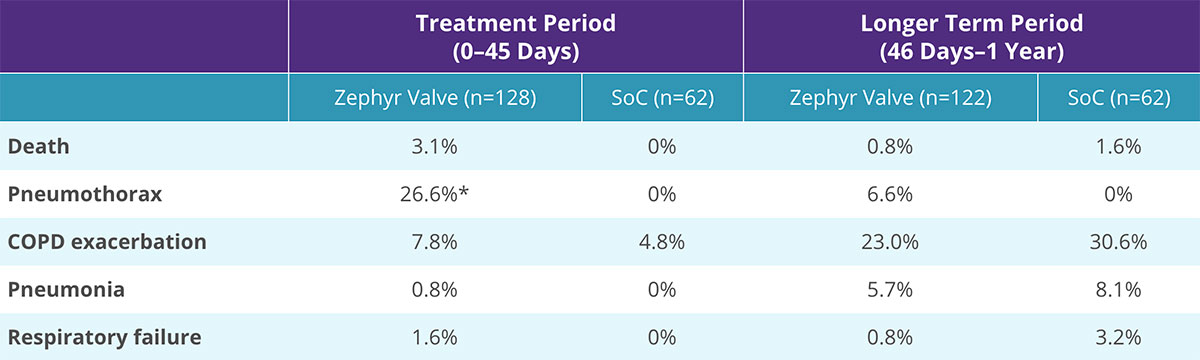
Safety: Pulmonary Serious Adverse Events (SAEs) Occurring in at Least 3.0% of Subjects in Either Group
- Increased SAE rate with Zephyr Valve treatment compared to SoC in the short-term (first 45 days post-procedure)
- Reduced SAE rate long-term (46 days to 12 months) with Zephyr Valve treatment compared to standard of care
- 5 of the 8 subjects experiencing a pneumothorax in the longer-term period had recently undergone a secondary bronchoscopy for valve replacement and/or removal
Conclusion
Zephyr® Endobronchial Valve treatment in carefully selected patients with little or no collateral ventilation in the target lobe provides clinically meaningful and statistically significant benefits in lung function, exercise tolerance, dyspnea, and quality of life over current standard of care medical therapy out to at least 12 months.
Complications of the Zephyr Endobronchial Valve treatment can include but are not limited to pneumothorax, worsening of COPD symptoms, hemoptysis, pneumonia, dyspnea and, in rare cases, death.