There’s Another Option for Your Severe COPD/ Emphysema Patients
See Why Pulmonologists are Choosing Zephyr® Valve to Reduce Hyperinflation
Changing the Standard of Care for Severe COPD/Emphysema Treatment:
- Gold Evidence “A” rating
- Received FDA approval in 2018
- Backed by 100 scientific studies
- >25k patients treated globally
- Precise patient selection tools
- Included in treatment guidelines

Interested in offering a full spectrum of treatment options for severe COPD/Emphysema?
Sign-up to receive information on endobronchial valves including patient selection, clinical case studies, and invitations to our latest webinars.
Clinical Benefits and Risks Observed in Patients Treated with Zephyr Valves
Clinically Significant Improvement in Lung Function1

Improved Quality of Life1

Improved Exercise Capacity1
Complications can include but are not limited to pneumothorax (tear in the lung), worsening of COPD symptoms, hemoptysis, pneumonia, and, in rare cases, death.
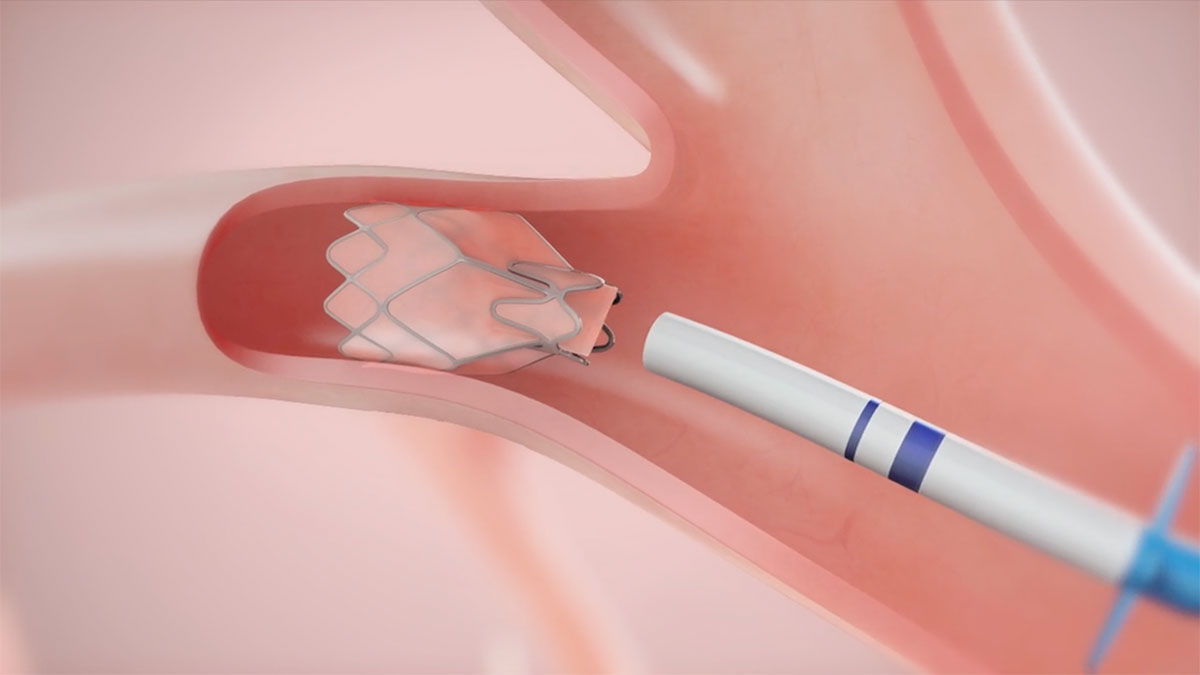
See the Zephyr Valve Mechanism of Action
The Zephyr Endobronchial Valve is indicated for the bronchoscopic treatment of patients with hyperinflation associated with severe emphysema in regions of the lung that have little to no collateral ventilation (CV).
You can use this minimally invasive, implantable device to occlude all airways feeding the hyperinflated lobe of a lung that is most diseased.
Watch this video to see how these unique one-way valves reduce gas trapping.

Your Patients May Qualify
Patients who meet the following criteria should be evaluated for treatment with endobronchial valves:1–5
- Breathless despite optimal medical management (mMRC≥2)
- A confirmed diagnosis of COPD
- Non-smoking or willing to quit smoking
- Have an FEV1 ≤50% predicted


GOLD 2020: Level A Evidence rating affirms that endobronchial valves, like the Zephyr Valve, are a viable, minimally invasive treatment option for severe emphysema, a form of COPD.

© 2022 Pulmonx Corporation or its affiliates. All rights reserved. All trademarks herein are the property of Pulmonx Corporation or its affiliates. US-EN-1467-v1
Privacy Policy | Terms of Use | Sitemap | Important Safety Information | In The News | Contact Us