Welcome to the Virtual Tour for Interventional Pulmonologists
Breakthrough Technology for Severe Emphysema
Helping Patients Breathe Easier, Do More, and Enjoy Life1
The Zephyr® Valve delivers durable clinical benefits — without surgery and its associated risks.1-5
Patients experience:1-5
- Improved health status
- Improved lung function
- Increased exercise capacity
- Reduced breathlessness or dyspnea
- Reduced gas trapping
- Improved quality of life


Dr. Gerard J. Criner Takes You on Virtual Tour of a Zephyr Procedure*
Patient Case Study
Patient Information:
- 77-year-old female
- BMI 27
- FEV1 29% predicted
- TLC 156% predicted
- RV 228% predicted
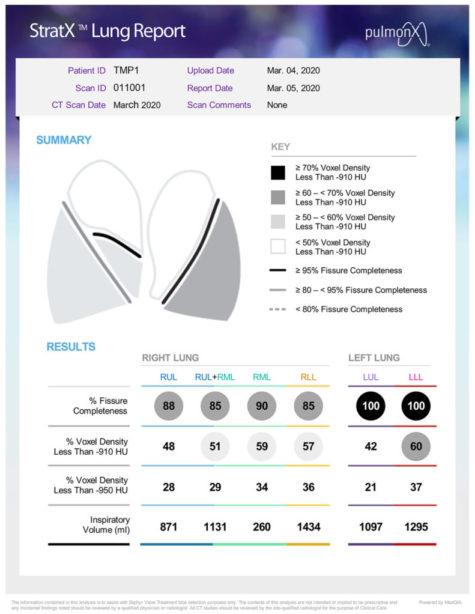
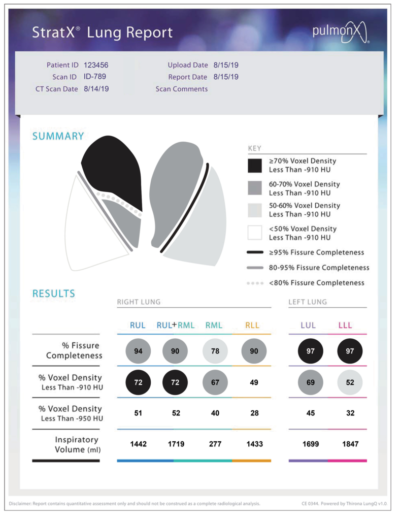
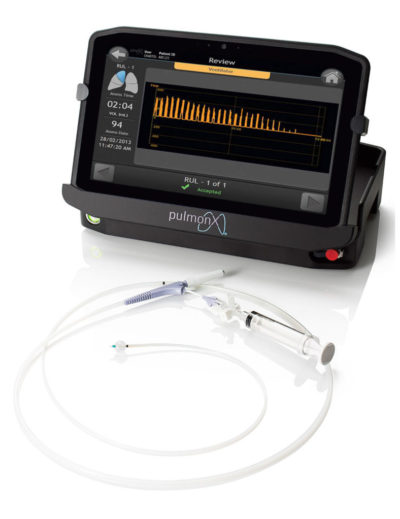
StratX® Platform
Prior to treatment, physicians perform a clinical workup, including lung function and hyperinflation testing. The StratX Lung Analysis Platform is a cloud-based quantitative CT analysis service that provides information on emphysema destruction, fissure completeness, and lobar volumes.
The StratX Lung Report is used in treatment planning to select primary, secondary, and tertiary target lobes for treatment. Dr. Criner used this StratX Lung Report to select this patient’s target lobe, verify fissure integrity and ultimately place 5 valves as seen in the below videos. This StratX report displays the left lower lobe as the most destructed (60%), with a high fissure completeness (100%) and high inspiratory volume (1,295).
Chartis® System Assesses Collateral Ventilation
The Chartis System has been validated in multiple randomized clinical trials1-4 to predict likely responders to the Zephyr Valve treatment. The Chartis System provides precise flow and pressure readings for specific lobes in the lung to assess absence of collateral ventilation (CV), which is a predictor of response to treatment.
In this video Dr. Criner demonstrates how the Chartis System is used to assess the presence of CV within isolated lobes of the lung.
Patient Overview:
- 77-year-old female
- BMI 27
- FEV1 29% predicted
- TLC 156% predicted
- RV 228% predicted
- 5 valves placed in the left lower lobe
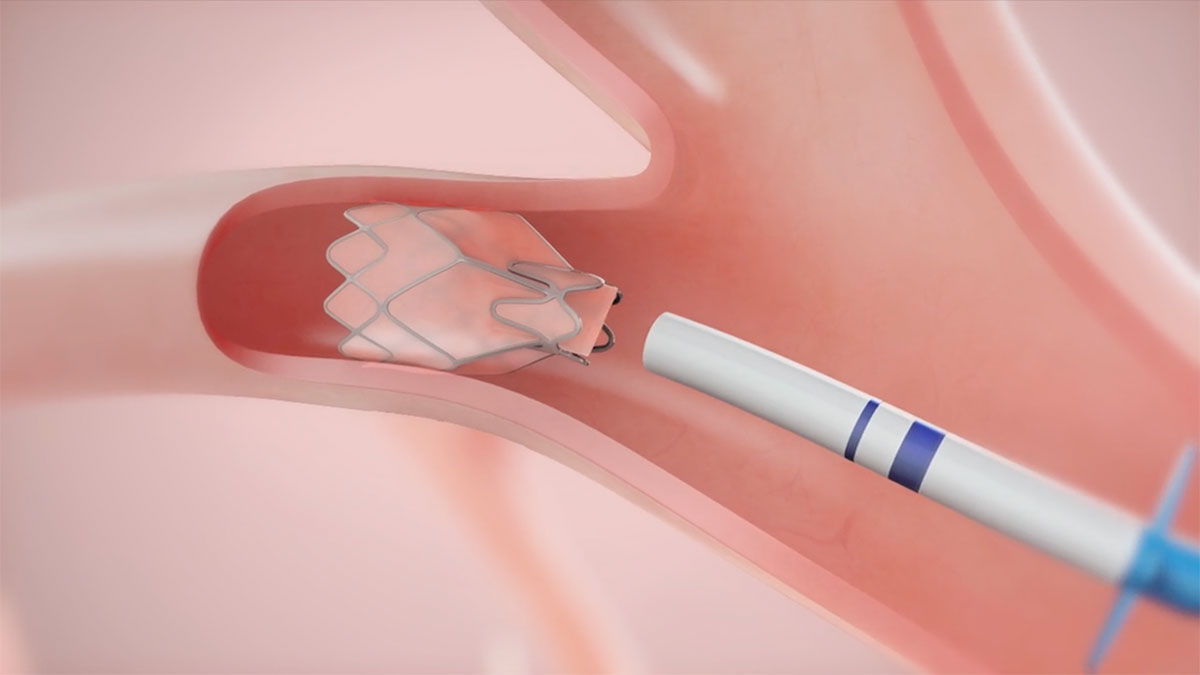
Zephyr Valve Placement
Dr. Criner demonstrates placing 5 Zephyr Valves in the Left Lower Lobe
Complications of the Zephyr Endobronchial Valve Treatment include but are not limited to pneumothorax, worsening of COPD symptoms, hemoptysis, pneumonia, dyspnea and, in rare cases, death.
Post Procedure Patient Outcomes
Chest CT Pre-Zephyr Procedure
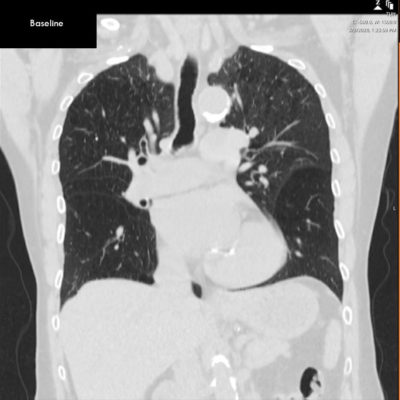
Baseline shows lower lobe predominant emphysema, hyperinflation and flattening of the diaphragm.
Chest CT Post-Zephyr Procedure
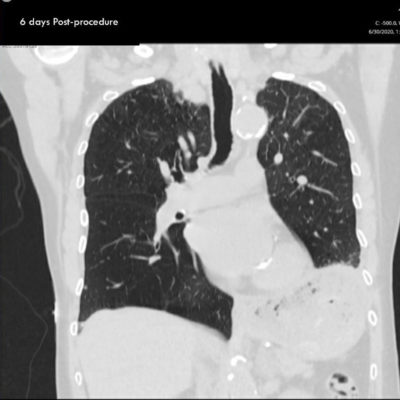
Post scan shows atelectasis of the left lower lobe and lifting of the diaphragm.
*Dr. Criner is a paid consultant for Pulmonx Corporation.
Clinical Evidence
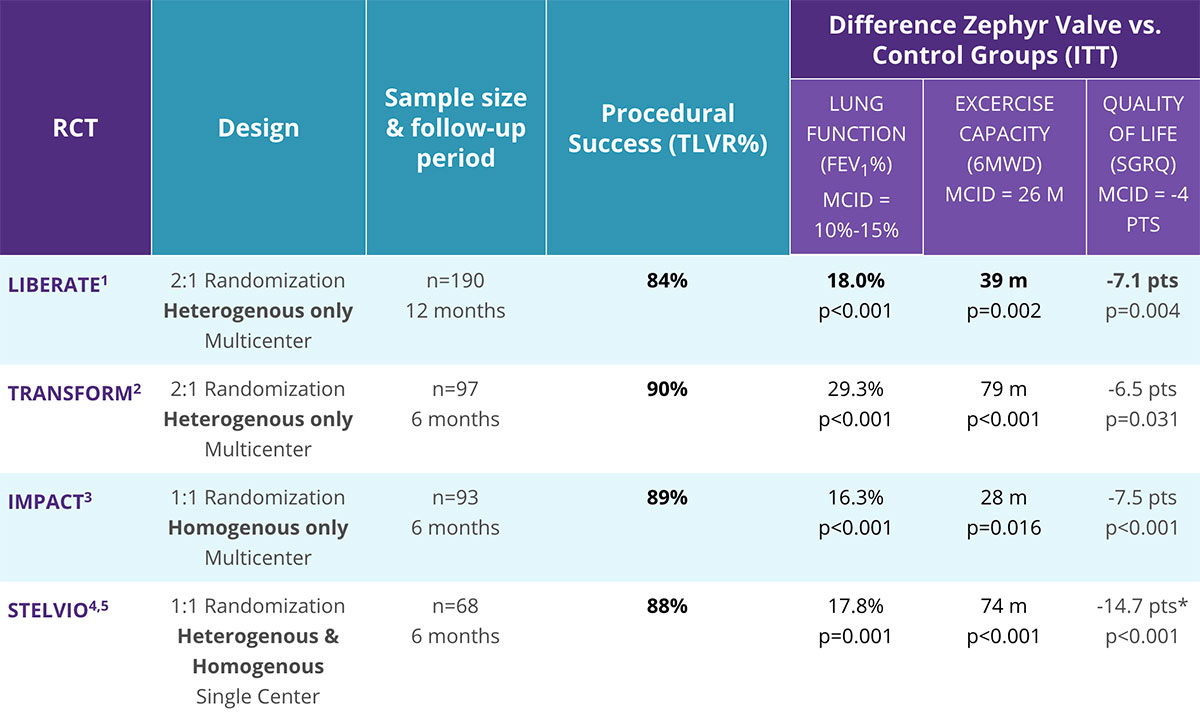
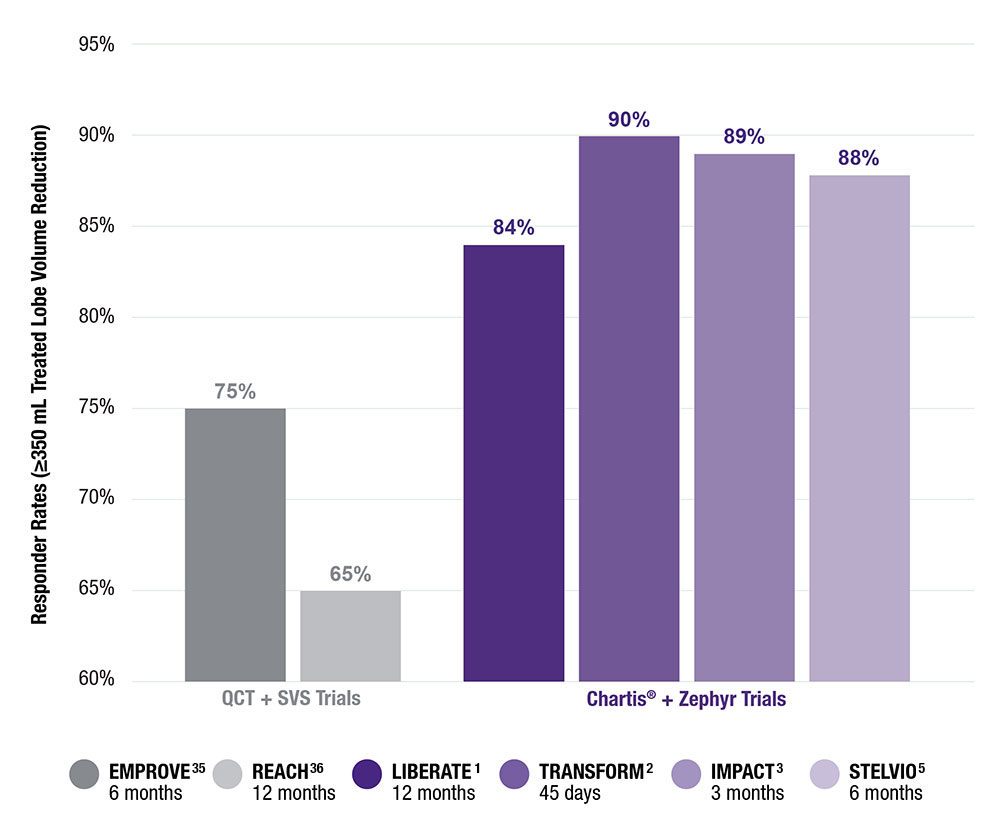
Consistent Clinical Findings Across Four Randomized Controlled Trials
Responder Rates for Zephyr Valve and Spiration Valve*
Responder Rates for Endobronchial Valves for Bronchoscopic Lung Volume Reduction1-4,35,36
Randomized Trials of CV- Patients**
The committee noted that there are different devices available for this procedure, and that the published evidence shows they may have different efficacy profiles.”
— NICE 201711
*Comparisons based on retrospective review of data, no head-to-head trials have been conducted.
**Collateral ventilation status determined by fissure integrity (QCT) or physiologic measure (Chartis) of CV.
Responder Rates and Incidence of Significant Pneumothorax
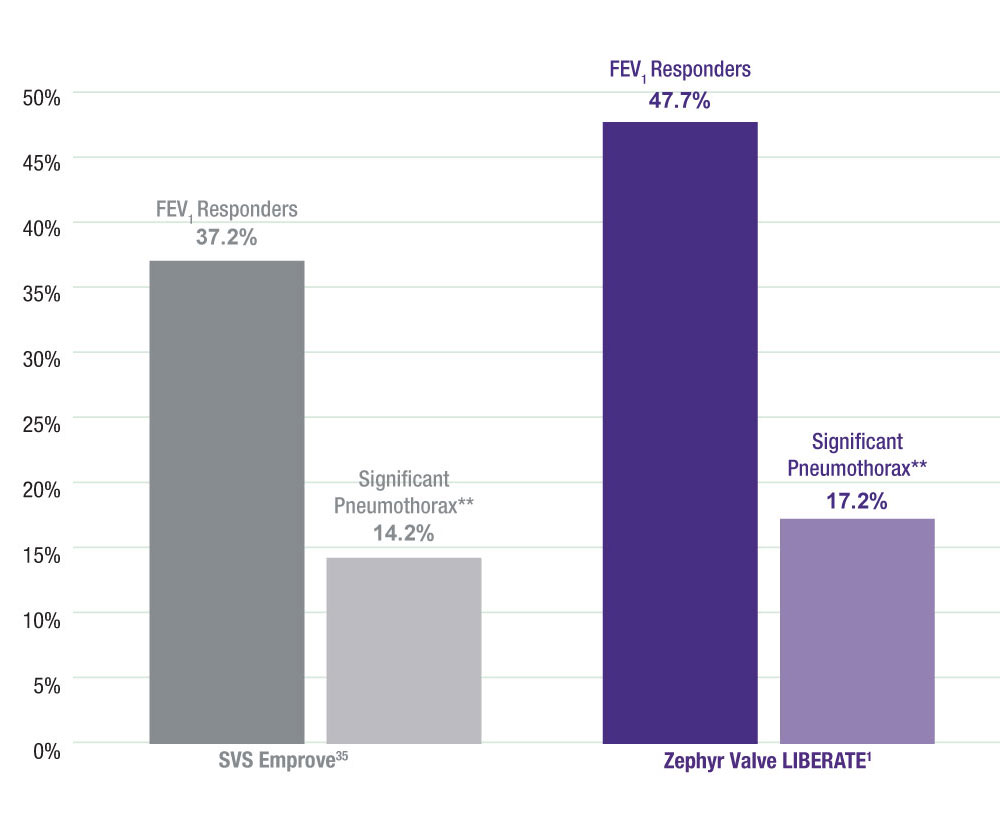
**Significant pneumothorax defined as a tension pneumo or a pneumo that requires >7 days hospitalization, surgery, all valve removal, or results in death
Pulmonologist Perspective:
How Zephyr Valves Work
Assess with Certainty. Treat with Confidence.
Predict successful TLVR with 90% accuracy.6
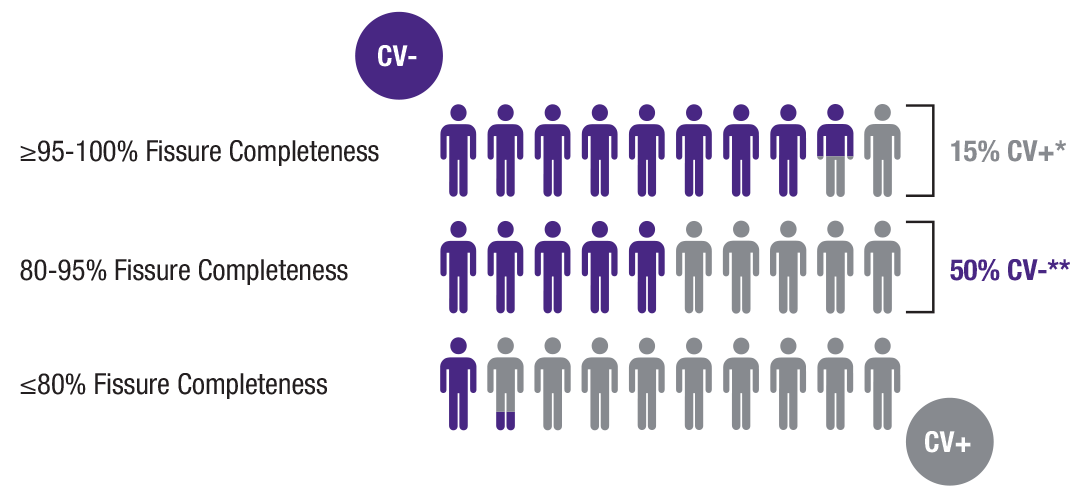
Patients without collateral ventilation (CV-) are more likely to respond to treatment with endobronchial valves. Pulmonx offers precise patient selection tools to help physicians identify those most likely to benefit from treatment.
StratX® Lung Analysis Platform
QCT Analysis:
Non-invasive to rule out non-responders
Chartis System Pulmonary Assessment System
Physiological assessment for collateral ventilation to identify likely responders
Using QCT alone may exclude patients with no collateral ventilation from being treated or treat patients with collateral ventilation who may receive no benefit.
Used prior to placement of valves, StratX Platform and the Chartis System give physicians confidence in their treatment decision and are a critical addition to analysis of fissure completeness. Without the Chartis System, physicians may treat a lobe that has collateral ventilation, or unnecessarily rule out a patient who could have benefitted.
Patient Selection Benefit Of Chartis System7
*Includes patients with “low plateau” measurements, typically not achieving TLVR when treated
**Includes patients with “low flow” readings in the target lobe, typically resulting in TLVR when treated
How It Works
See how the Zephyr Valve treatment is performed as a minimally invasive lung volume reduction procedure to reduce hyperinflation and gas trapping in patients with severe emphysema. The Zephyr Valve is the first endobronchial valve to receive approval from the FDA for patients with either heterogenous or homogeneous emphysema.
Comprehensive Solutions to Support
You and Your Patients
Pulmonx is committed to serving hospitals, physicians, and patients across the continuum of care to help you deliver superior patient outcomes.

Professional Education & Case Support
Pulmonx delivers high quality professional education for physicians and their teams to help them achieve superior patient outcomes and patient satisfaction.
- Off-site dedicated training sessions with the field’s leading experts for treating physicians and their teams
- On-demand comprehensive training module
available online - Patient selection case study training modules

Patient Access & Reimbursement Support
Pulmonx provides dedicated support to healthcare providers and associated facilities, patients, and their families as they navigate through the reimbursement process.
- Supporting patients and providers as they navigate coverage with a dedicated team of experienced
case managers - Collaborating with your administrative and coding and billing teams to ensure appropriate understanding of reimbursement situation
- Educating payers and monitoring insurance coverage developments impacting patient access

Research & Development
Our team is developing leading edge tools to deliver superior patient outcomes.
- Gaining insights from physicians and through evidence-based medicine to enhance products and assessment tools
- Investing in long-term clinical research with meaningful primary and secondary endpoints

Patient Awareness & Market Development Support
Many patients living with emphysema don’t know that they have options. Pulmonx is dedicated to increasing awareness of this treatment.
- Engagement with patients and patient advocates
- Promotion of a patient-centered website with a Zephyr Treatment Center locator
- Partner and sponsor of events for patient awareness
- Educational resources that treating facilities and professionals may use for community outreach
Chat with Us
Featured Patient Stories

Charlie’s Story
Age at Treatment: 68
Fairfax, Virginia
My story about this procedure tells people what life can be even with COPD. It really brought me back. I am a walking Christmas present.


Flo’s Story
Age at Treatment: 63
Texas City, TX
I feel strongly that part of my new path in life is to help spread the word about the Zephyr Valves so other sufferers can learn about this option and have hope.